Underground natural gas storage in salt caverns and the challenge of hydrate formation
Underground natural gas storage
Natural gas remains one of the most important energy sources in the world despite the increasing popularity of renewable energy. In terms of emissions during power generation, natural gas is the cleanest fossil fuel. According to the United States Energy Information Administration (EIA), use of natural gas produces almost 50% less CO2 than coal.
In order, to ensure a reliable, year-round supply of affordable natural gas, large storage facilities are required. While natural gas can be stored in gaseous and liquified form in above ground tanks, underground storage offers several benefits including economy and safety. Underground natural gas storage facilities can be classified into three main types.
For the curious minded reader, a brief overview of the different types of underground natural gas storage facilities is given in the info box at the end of the article. This article only focuses on natural gas storage in salt caverns.
How is natural gas stored in salt caverns?
Salt is impermeable to natural gas. It is this property that makes salt caverns ideal for natural gas storage.
A natural gas cavern is a cavity “dug” in a salt dome located 1000 to 2500m below the earth’s surface. The first step in creating a salt cavern is to find a salt deposit location suitable for natural gas storage. The solubility of salt in water is used to remove the salt from the dome thus creating a cavity.
A well is drilled in the salt formation and a large amount of water is cycled through the completed well. The water dissolves the salt increasing the size of the cavity. The salty water extracted from the well is further processed to remove the salt or released into the sea in an environment-friendly way.
This process of creating a salt cavern is called ‘salt cavern leaching’. The leaching process may take several years until a sufficiently large cylindrical cavity has formed.
Salt caverns typically have a diameter of up to 100m and a height of 500m; this is large enough to house the Berlin television tower!
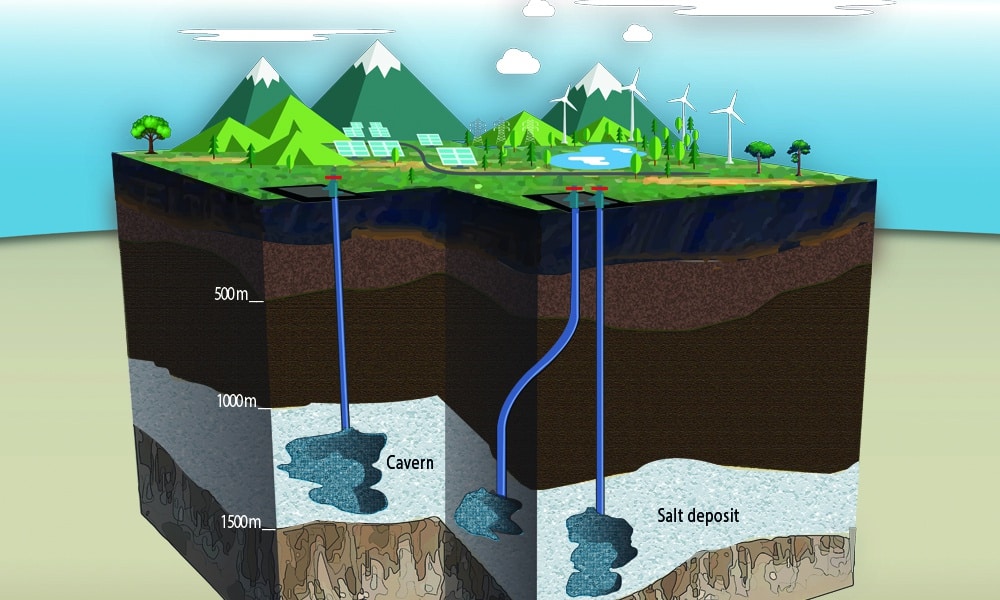
Salt cavern leaching can be relatively expensive when compared to the cost of setting up other types of underground natural gas storage. However, the deliverability of a salt cavern is typically much higher than for an aquifer or a depleted reservoir.
Deliverability is the amount of gas that can be withdrawn from a storage facility per day. High deliverability not only means that stored natural gas can be more readily withdrawn, but also that it can be replenished faster.
Furthermore, the cushion gas requirement for a salt cavern is the lowest of the three storage types. Salt caverns typically require only a third of the total gas capacity to be used as cushion gas.
Along with these advantages, storage of natural gas in salt caverns also presents challenges. A major issue faced by operators of natural gas storage facilities is gas hydrate formation in the piping. Or in equipment connecting the reservoir head to the treatment plant, too. During withdrawal or supply of gas from/to a salt cavern, problems such as restricted flow and inaccurate pressure measurements can arise due to the formation of hydrates.
What are hydrates?
Methane clathrate, also known as natural gas hydrate or methane hydrate is a solid clathrate compound in which a large amount of methane is trapped.
Clathrate refers to a substance that traps molecules inside a lattice. In this case, the lattice is formed by water molecules held together by hydrogen bonds.
How does hydrate formation occur?
The four essential components that lead to hydrate formation during the transport of natural gas from the reservoir are:
- Water
- Light hydrocarbon gases
- Low temperature
- High pressure
Low temperature and high pressure:
The conditions of low temperature and high pressure are generally met inside pipelines especially in cold areas and subsea applications.
Water and light hydrocarbon gases:
Brine water in salt caverns is drained as gas is filled into the cavern. The residual water from the process can form hydrates when natural gas is withdrawn.
Hydrate formation can occur during normal production, but transient operations are often more susceptible.
What are the problems related to hydrate formation?
Natural gas hydrates consist of approximately 85 mole percent water. The physical properties of hydrates are, therefore, similar to those of ice. Hydrates typically float at the water/hydrocarbon interface. They can agglomerate into bigger clusters and plug valves, pumps, and other narrow conduits or even the whole pipeline. The ice-like solid hydrates reduce the transmission capacity and may over time, block and possibly rupture the pipeline.
In large transport pipelines, hydrate formation is countered by removing moisture from the gas using additives such as glycerin. This approach, however, does not work with measuring lines that do not have product flow.
Hydrates can clog these lines and impair the function of measuring sensors and instruments. As a result, pressure and differential pressure readings can be erroneous; in extreme cases, it may even become impossible to obtain any measurements at all.

Conclusion
Storage of natural gas in underground salt caverns offers several advantages. For example a high deliverability, low cushion gas requirement, and a long service life.
A major challenge, however, during use of such storage facilities is the formation of hydrates in the supply and measurement lines. Hydrate formation can obstruct gas flow reducing supply which can lead to pipeline rupture.
Furthermore, clogging of measurement lines can make it difficult to obtain accurate pressure measurements.
INFO-BOX
Types of underground natural gas storage
An underground natural gas storage facility acts as a very large storage vessel. Natural gas can be injected into the underground formation, building up pressure. Comparable to a pressurized natural gas container.
As stored gas is consumed, the pressure drops below that of the wellhead. Thus, there is no longer a pressure differential to push the gas out of the reservoir.
Underground storage facilities, therefore, have a certain amount of gas that is not recoverable. The amount of gas permanently stored in a natural gas storage is called cushion gas. The amount of cushion gas required depends on the type of storage and the desired withdrawal rate.
Underground natural gas storage facilities can be classified into three main types:
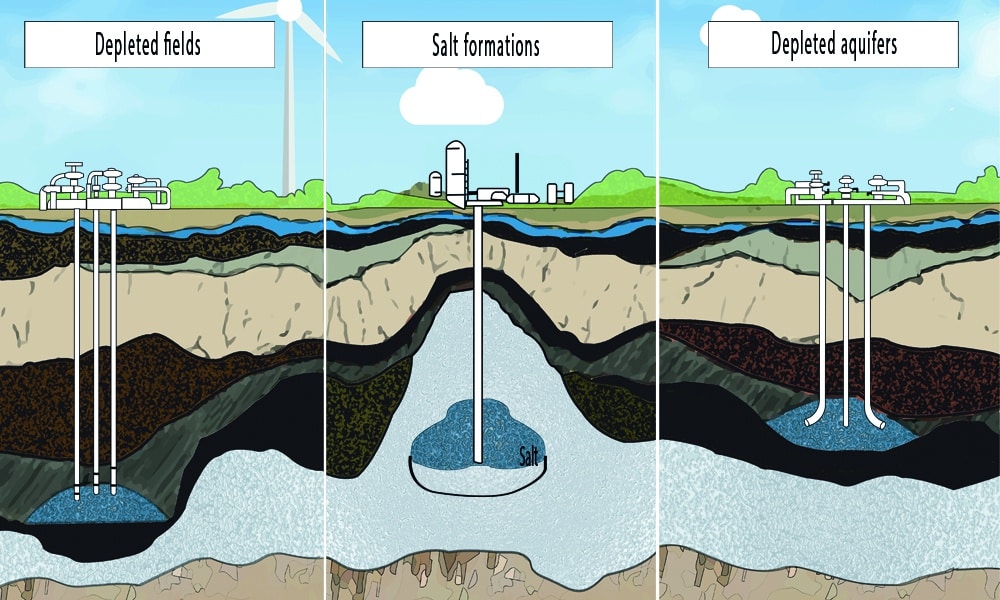
Depleted gas reservoirs
Depleted gas reservoirs are the most common form of underground gas storage. Natural gas reservoirs that have been tapped of most of their recoverable natural gas can be converted into gas storage facilities. These underground formations have proven geological capability of holding natural gas. And can, therefore, serve as gas storage sites. In depleted gas reservoirs, around half of the total volume consists of cushion gas.
Depleted aquifers
Aquifers are underground permeable rock formations that act as water reservoirs. In certain cases, these water-bearing formations can be repurposed for use as natural gas storage facilities. Aquifers, however, are more expensive to develop than depleted gas reservoirs. This type of storage is generally used in areas where there are no depleted reservoirs nearby. Aquifer reservoirs require up to 80% of the total volume as cushion gas.
Salt caverns
Salt caverns, created in underground salt formations, are another option for natural gas storage. The walls of a salt cavern are structurally as strong as steel. That make the reservoir highly resistant to degradation over the life of the facility. Salt caverns typically require around 33% of the total volume of the cavern as cushion gas.
Conclusion
Storage of natural gas in underground salt caverns offers several advantages such as high deliverability, low cushion gas requirement, and a long service life. A major challenge, however, during use of such storage facilities is the formation of hydrates in the supply and measurement lines. Hydrate formation can obstruct gas flow reducing supply which can lead to pipeline rupture. Furthermore, clogging of measurement lines can make it difficult to obtain accurate pressure measurements.
For information about how formation of hydrates in measurement lines can be prevented, please refer to the following case study:
Image Source: © matrivgraphics (Fiverr) & AS-Schneider

